Method Verification of Carbon and Sulfur Determination in Ferronickel Tapping Samples Using HCS-801 in Accordance with GB/T 20123
Main Article Content
Abstract
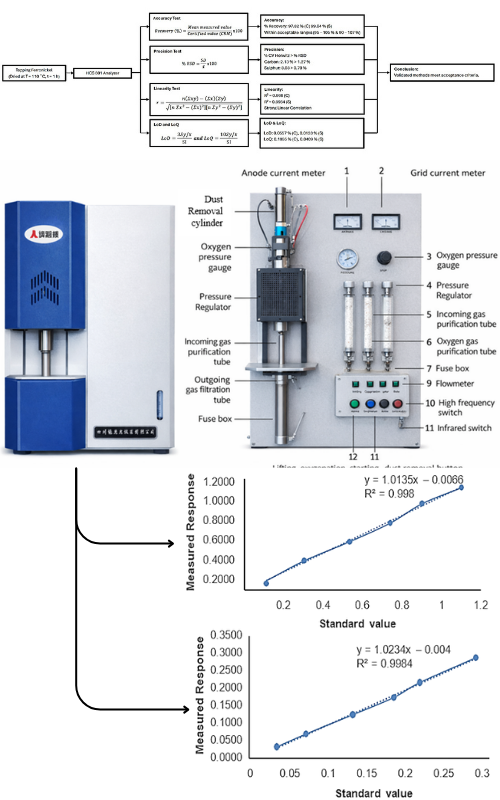
Reliable determination of carbon (C) and sulfur (S) in tapping ferronickel is critical for metallurgical quality control and compliance with industrial specifications. Given the complex metallic matrix of ferronickel, verification of analytical performance is essential before routine application. This study verifies the GB/T 20123 standard method for the simultaneous determination of carbon and sulfur in tapping ferronickel using a High Carbon Sulfur (HCS-801) combustion–infrared analyzer. Method performance was assessed using accuracy, precision (repeatability), linearity, limit of detection (LOD), and limit of quantification (LOQ), in accordance with internationally accepted validation principles. Evaluation of recovery testing yielded 97.82 % for carbon and 99.04 % for sulfur, both within acceptable recovery ranges. Precision expressed as %RSD was 1.27% for carbon and 0.70% for sulfur, complying with the Horwitz criterion (% RSD < ½ CV Horwitz) and the general acceptance limit of 2 %. Excellent linearity was obtained across the calibration range, with correlation coefficients (r) of 0.998 for carbon and 0.9984 for sulfur. LOD and LOQ values, calculated based on the standard deviation of response and slope of calibration curves, were 0.0557% and 0.1855% for carbon, and 0.0123 % and 0.0409 % for sulfur, respectively. The results demonstrate that the GB/T 20123 method, when implemented with the HCS-801 analyzer, provides reliable analytical performance for carbon and sulfur determination in tapped ferronickel and is suitable for routine industrial applications.
Downloads
Article Details
Section

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
How to Cite
References
[1] Y. Wang, A. Karasev, J. H. Park, and P. G. Jönsson, “Non-metallic Inclusions in Different Ferroalloys and Their Effect on the Steel Quality: A Review,” Metallurgical and Materials Transactions B: Process Metallurgy and Materials Processing Science, vol. 52, no. 5, pp. 2892–2925, Oct. 2021, doi: https://doi.org/10.1007/s11663-021-02259-7.
[2] Y. Han and M. Tangstad, “Metallurgical Properties of Biocarbon in Ferroalloy Production─A Review,” Jun. 11, 2024, American Chemical Society. doi: https://doi.org/10.1021/acsomega.4c00866.
[3] M. R. Bayati and Pengyi WEI, “Study on Carbon and Sulfur Detection by Using Infrared Absorption,” Adv. Mat. Res., vol. 989–994, pp. 146–150, 2014. doi: https://doi.org/10.4028/www.scientific.net/AMR.989-994.146.
[4] Gui Biao Tui Jian (GB/T), “Testing Standards GBT 20123,” 2006. [Online]. Available: https://www.chinesestandard.net/PDF.aspx/GBT20123-2006.
[5] J. Pum, “A practical guide to validation and verification of analytical methods in the clinical laboratory,” in Advances in Clinical Chemistry, vol. 90, Academic Press Inc., 2019, pp. 215–281. doi: https://doi.org/10.1016/bs.acc.2019.01.006.
[6] I. Ramadhan and R. Ahdiaty, “Verification of the COD Test Method by UV-Vis Spectrophotometry for Low and High Concentrations,” Ind. J. Chem. Anal, vol. 05, no. 01, pp. 52–61, 2022, doi: https://doi.org/10.20885/ijca.vol5.iss1.art6.
[7] B. N. Kumar, S. Kanchi, M. I. Sabela, K. Bisetty, and N. V. V. Jyothi, “Spectrophotometric determination of nickel (II) in waters and soils: Novel chelating agents and their biological applications supported by DFT method,” Karbala International Journal of Modern Science, vol. 2, no. 4, pp. 239–250, Dec. 2016, doi: https://doi.org/10.1016/j.kijoms.2016.08.003.
[8] M. M. Hatat-Fraile and B. Barbeau, “Performance of colorimetric methods for the analysis of low levels of manganese in water,” Talanta, vol. 194, pp. 786–794, Mar. 2019, doi: https://doi.org/10.1016/j.talanta.2018.11.003.
[9] A. Jena GmbH, “Determination of Total Carbon and Total Sulfur Content in Cement and (EN),” 2023. [Online]. Available: https://www.analytik-jena.com/import/assets/12720862_AppNote_EA_0042_TC_TS_cement_EN.pdf.
[10] A. S. Camera, P. P. Arcênio, W. de O. Pacheco Filho, T. de A. Maranhão, F. J. S. de Oliveira, and V. L. A. Frescura, “Method development and validation for sulfur determination via CS molecule in petroleum green coke by high resolution continuum source molecular absorption spectrometry,” Microchemical Journal, vol. 134, pp. 301–308, Sep. 2017, doi: https://doi.org/10.1016/j.microc.2017.06.022.
[11] M. L. Astolfi et al., “A multi-analytical approach to studying the chemical composition of typical carbon sink samples,” Sci. Rep., vol. 13, no. 1, Dec. 2023, doi: https://doi.org/10.1038/s41598-023-35180-x.
[12] F. Sruya Jaya and A. Tri Prasetya, “Validation of the Analytical Method for Cadmium (Cd) in Rice (Oryza Sativa) using the SNI 6989-84:2019”, ijcs, vol. 14, no. 2, Aug. 2025, doi: https://doi.org/10.15294/ijcs.v14i2.20607.
[13] B. M. Marson, V. Concentino, A. M. Junkert, M. M. Fachi, R. O. Vilhena, and R. Pontarolo, “Validation Of Analytical Methods In A Pharmaceutical Quality System: An Overview Focused On Hplc Methods,” Quim. Nova, vol. 43, no. 8, pp. 1190–1203, Sep. 2020, doi: https://doi.org/10.21577/0100-4042.20170589.
[14] P. Chanda Gupta, “Method Validation of Analytical Procedures,” Pharmatutor, vol. 3, no. 1, pp. 32–39, 2015. [Online]. Available: https://www.pharmatutor.org/magazines/articles/january-2015/method-validation-analytical-procedures.
[15] T. Nageswara Rao, “Validation of Analytical Methods,” in Calibration and Validation of Analytical Methods - A Sampling of Current Approaches, M. T. Stauffer, Ed., London: IntechOpen, 2018. doi: https://doi.org/10.5772/intechopen.72087.
[16] S. Goulikar and T. P. Raj, “Development And Validation of New Analytical Method for the Estimation Of Flupentixol Dihydrochloride in Pure And Pharmaceutical Dosage Form,” Certified Journal │ 986 World Journal of Pharmaceutical Research SJIF Impact Factor 8, vol. 11, no. 5, pp. 986–1048, 2022, doi: https://doi.org/10.20959/wjpr20225-23984.
[17] D. A. Afifah, M. Muslihudin, and D. Cendekia, “Quality Control Implementation for Accuration and Precision of Protein Analysis,” 2021. doi: https://doi.org/10.33024/jaf.v7i1.5444.
[18] M. Pratiwi and M. I. Syawal, “Verifikasi Logam Berat K (Kalium) dengan Metode Flame Spektrofotometri Serapan Atom (SSA),” Asian Journal of Science, Technology, Engineering, and Art, vol. 1, no. 2, pp. 229–242, Oct. 2023, doi: https://doi.org/10.58578/ajstea.v1i2.2001.
[19] H. Y. Chen and C. Chen, “Evaluation of Calibration Equations by Using Regression Analysis: An Example of Chemical Analysis,” Sensors, vol. 22, no. 2, Jan. 2022, doi: https://doi.org/10.3390/s22020447.
[20] A. Dwi Nanda et al., “Indonesian Journal of Chemical Science Validation of Methods and Determination of Curcumin Levels in Carrying Herbal Medicine of Turmeric Tamarind Using UV-Vis Spectrophotometer,” J. Chem. Sci, vol. 14, no. 2, 2025, doi: https://doi.org/10.15294/ijcs.v14i2.14192.
[21] S. Sugito, and A. K. Rachmad Setiawan, " AAS Thermo Ice 3000 Performance Test on Cu Metal Using CRM 500 and CRM 697 at the UNS Integrated Laboratory UPT," Jurnal Pengelolaan Laboratorium Pendidikan, vol. 4, no. 1, pp. 1-6, Jan. 2022. https://doi.org/10.14710/jplp.4.1.1-6.
[22] B. M. Gandhi, A. L. Rao, and J. V. Rao, “Method Development and Validation for Simultaneous Estimation of Montelukast Sodium and Desloratadine by RP-HPLC,” Am. J. Analyt. Chem., vol. 06, no. 08, pp. 651–658, 2015, doi: https://doi.org/10.4236/ajac.2015.68063.
[23] N. W. Lin Wang, “Determination of carbon and sulfur in copper-lead-zinc polymetallic ores by high-frequency combustion infrared absorption method,” Chinese Journal of Inorganic Analytical Chemistry, vol. 14, no. 11, 2024. doi: https://doi.org/10.13228/j.boyuan.issn1000-7571.140169.
[24] Harmita, “Instructions for Implementing Method Validation and Calculation Methods,” Majalah Ilmu Kefarmasian, vol. 1, no. 3, pp. 117–135, 2004. [Online]. Available: https://scholarhub.ui.ac.id/mik/vol1/iss1/12/.
[25] AOAC International, “Appendix F- Guidelines for Standard Method Performance Requirements,” Official Methods of Analysis, 2016. [Online]. Available: https://www.aoac.org/wp-content/uploads/2019/08/app_f.pdf.
[26] D. Cahyadi et al., “Method Validation for Determination of Calcium, Magnesium, and Zinc Content in Lubricant by Atomic Absorption Spectrometry (AAS) using Direct Dilution Technique by Xylene,” 2020. doi: https://doi.org/10.31153/js.v23i1.823.
[27] T. Aprianto, R. Hadiyati Noor, D. Analis Kimia, and F. “Verification of the Method for Determining Acetosal In Headache Medication Using The Uv Spectrophotometric Method,” Jurnal Sains dan Teknologi, Vol. 6, no. 1, Apr. 2017. doi: https://doi.org/10.23887/jstundiksha.v6i1.9398.
[28] W. Khoirul Umar, F. Agustiyar, and A. Rahma, “Verification of the Walkley Black Method Test for Determination of Organic Carbon Elements in Palm Oil Empty Fruit Bunch Waste Fertilizer,” Eksergi, Vol. 19, no. 3, Nov. 2022. doi: https://doi.org/10.31315/e.v19i3.7267.
[29] I. Solihat, A. P. Tirta, A. P. Ramdani, and A. Nandang Roziafanto, “Verification of the Testing Method for Nitrite Levels in Wastewater Using UV-Visible Spectrophotometry,” KOVALEN: Jurnal Riset Kimia, vol. 8, no. 1, pp. 53–59, Apr. 2022, doi: https://doi.org/10.22487/kovalen.2022.v8.i1.15756.